This article helps healthcare readers compare octenidine and chlorhexidine sources without turning a study, ingredient name, guideline mention, or product label into a product recommendation.
It is not a protocol, substitution guide, procurement recommendation, dosing reference, patient-selection tool, or statement that either antiseptic is generally better. A local decision needs the current product label, local protocol, patient context, infection prevention review, and qualified clinical judgment.
Quick Position
The safest comparison is the narrow one: which product, in which setting, against which comparator, for which endpoint, in which jurisdiction.
Chlorhexidine has a larger guideline footprint in several U.S. and UK healthcare contexts. CDC catheter guidance names chlorhexidine-containing approaches for patient cleansing, catheter insertion skin antisepsis, and selected catheter-related products. NICE surgical-site guidance lists alcohol-based chlorhexidine as the first skin-preparation option unless contraindicated or the surgical site is next to a mucous membrane, and WHO surgical-site guidance includes chlorhexidine-containing alcohol-based solutions in its global guideline context.
Octenidine has clinical evidence in selected settings, especially European healthcare-associated infection, decolonization, bathing, wound, and product-specific contexts. A 2023 clinical review found 31 clinical articles through August 21, 2022, but it also found heterogeneous decolonization results, weak evidence for some preoperative washing bundles, no direct study comparing octenidine for pre-incisional surgical skin preparation with other antiseptics, and mostly negative evidence for daily octenidine bathing reducing ICU- or catheter-related bloodstream infections.
Later ICU evidence makes the comparison more nuanced, not simpler. A 2023 CLIP-ID post-hoc analysis found lower ICU-attributable CLABSI after implementation of chlorhexidine bathing but not after octenidine bathing. A 2024 German EFFECT trial found that octenidine-impregnated washcloths reduced ICU-acquired primary bacteremia but did not reduce ICU-acquired multidrug-resistant organism acquisition. Those studies used different designs, products, endpoints, and analytic frames.
Why Simple Comparisons Mislead
Octenidine and chlorhexidine are often discussed as if they were single interchangeable choices. In healthcare evidence, they usually appear as finished products or components of bundles.
For chlorhexidine, a source may be discussing chlorhexidine gluconate in alcohol, a chlorhexidine wash, a chlorhexidine-impregnated dressing, or another finished product. The DailyMed ChloraPrep label, for example, identifies a chlorhexidine gluconate and isopropyl alcohol product for patient preoperative skin preparation and includes product-specific warnings. That label context cannot be applied to every chlorhexidine product.
For octenidine, a source may be discussing octenidine dihydrochloride, octenidine hydrochloride, an octenidine wash mitt, an octenidine-impregnated washcloth, an octenidine nasal product, or a combination product with phenoxyethanol. The FDA GSRS record lists octenidine hydrochloride and includes octenidine dihydrochloride as a synonym, while also stating that UNII availability does not imply regulatory review or approval. That identity record is not clinical evidence.
The comparison has to stay attached to product, formulation, body site, patient population, endpoint, and source type.
Surgical Skin Preparation
For surgical skin preparation, major guidelines more often name chlorhexidine than octenidine. NICE recommends preparing the skin immediately before incision with an antiseptic preparation and lists alcohol-based chlorhexidine as the first option unless contraindicated or the site is next to a mucous membrane. The same NICE section warns about risks of skin antiseptics in babies, especially severe chemical injuries with chlorhexidine in preterm babies, and says some products may not be licensed for surgical-site antiseptic skin preparation.
WHO describes its surgical-site infection guidelines as globally valid and suitable for local adaptation. Its surgical-site skin-preparation recommendation context should still be read as guideline-level infection prevention evidence, not as permission to substitute products across countries or labels.
The octenidine evidence base does not provide the same kind of direct pre-incisional comparator support. The 2023 octenidine clinical evidence review stated that no study compared octenidine for skin preparation before surgical interventions with other antiseptics. It found weak evidence for preoperative washing in orthopedic and cardiac surgery when octenidine was combined with other topical measures.
A surgical-prep comparison should therefore avoid class-wide claims that octenidine is better, equivalent, or inferior. Chlorhexidine appears directly in several guideline and label contexts, while octenidine surgical evidence is more limited and often bundled or indirect.
Catheter And ICU Bathing Evidence
Catheter-related sources require especially careful endpoint separation. CDC catheter guidance names chlorhexidine in several recommendations, including a 2% chlorhexidine wash for daily skin cleansing to reduce catheter-related bloodstream infection and a chlorhexidine preparation with alcohol for skin antisepsis during central venous catheter insertion. The 2022 CLABSI prevention update also lists daily chlorhexidine bathing for ICU patients older than 2 months and alcoholic chlorhexidine skin preparation among essential practices.
Those recommendations do not automatically answer whether an octenidine product should replace chlorhexidine in a local catheter bundle. They show that chlorhexidine has guideline support in that source set.
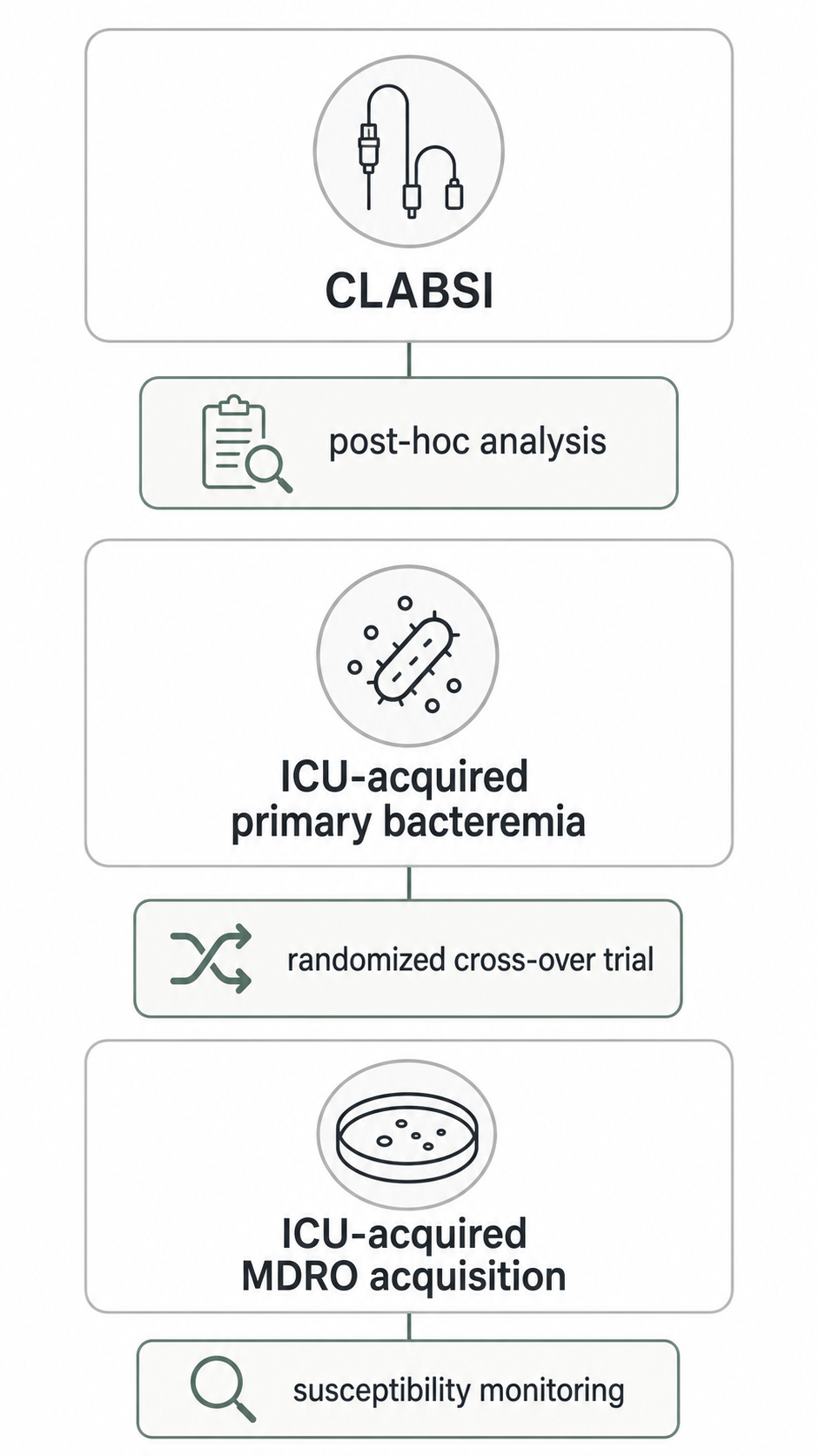
The 2023 CLIP-ID post-hoc analysis compared ICU-attributable CLABSI rates before and after implementation of daily bathing with 2% chlorhexidine-impregnated cloths, 0.08% octenidine wash mitts, or water and soap. It reported lower CLABSI incidence density in the chlorhexidine group, from 1.48 to 0.90 CLABSI per 1000 central-line days, and no reduction in the octenidine group, from 1.26 to 1.47 CLABSI per 1000 central-line days. The adjusted incidence rate ratio was 0.63 for chlorhexidine and 1.17 for octenidine.
The 2024 EFFECT trial asked a different question. It was a multicenter, cluster-randomized, double-blind, placebo-controlled, cross-over study in 44 ICUs in 23 German hospitals. It compared octenidine-impregnated washcloths with placebo washcloths and reported a lower hazard of ICU-acquired primary bacteremia, with no intervention effect for ICU-acquired multidrug-resistant organisms. A 2025 publisher correction changed one placebo-event count in a table from 51 to 351, and the original article has been corrected.
Those findings can coexist because the endpoint and design are not the same. CLABSI, ICU-acquired primary bacteremia, and ICU-acquired multidrug-resistant organism acquisition should not be collapsed into a single “ICU infection prevention” conclusion.
Staphylococcus Aureus Decolonization
For Staphylococcus aureus prevention in acute-care facilities, CDC describes strategies that include intranasal antistaphylococcal antibiotic or antiseptic options with chlorhexidine wash or wipes for selected high-risk periods and settings. That CDC page is a healthcare-facility strategy source, not an octenidine comparison source.
The 2023 octenidine clinical evidence review found that octenidine-containing decolonization studies were heterogeneous. Decolonization success ranged widely, and many studies involved bundles, different carrier definitions, different follow-up windows, or co-interventions.
One review-identified study assessed octenidine versus chlorhexidine bathing in a methicillin-resistant Staphylococcus aureus carriage context and found reductions in carriage prevalence for both compared with no antiseptic bathing. That finding is not enough to declare the agents interchangeable. It remains tied to the study setting, product selection, nasal treatment context, screening method, and endpoint.
Susceptibility Monitoring Is A Separate Question
Clinical effectiveness and antiseptic susceptibility are related but different questions. The PLOS ONE analysis from the CLIP-ID trial assessed bacterial isolates collected before and after routine ICU bathing with chlorhexidine, octenidine, or water and soap. It found no evidence of reduced phenotypic susceptibility to chlorhexidine or octenidine after 12 months in the studied ICU isolates.
The same paper cautioned that the observation period might be too short and species-level sample sizes too small for final conclusions. That is an important limit. The result should not be rewritten as “no resistance risk,” “safe for long-term use,” or “no need for surveillance.”
Wound And Mucosal Contexts
Wound and mucosal comparisons are more product-specific than many summaries suggest. Octenidine evidence often involves wound or mucosal products, and some products include phenoxyethanol. Chlorhexidine evidence often involves skin, bathing, catheter, or surgical-prep products. Neither source family should be moved into wounds, mucosa, eyes, ears, burns, deep wounds, neonatal care, pregnancy, or device care without exact label and protocol review.
For wound-care context, see Octenidine In Wound Care Evidence. That article keeps wound type, comparator, product, and endpoint separate and does not provide wound-care instructions.
How To Write A Responsible Comparator Claim
A responsible healthcare comparison should usually use narrow wording:
- In this guideline, chlorhexidine is named for this specific infection prevention practice.
- In this trial, this octenidine product was studied for this endpoint in this setting.
- In this review, the evidence for octenidine was mixed, heterogeneous, or limited for this context.
- This label supports product-specific composition, intended use, and warnings, not broad comparative effectiveness.
- This identity record confirms a substance name or synonym, not clinical authorization or product interchangeability.
Avoid broad wording:
- Octenidine is better than chlorhexidine.
- Chlorhexidine is always preferred.
- Octenidine can replace chlorhexidine.
- Both agents are equivalent.
- A positive ICU bathing result applies to surgical prep, wound care, mucosa, or device care.
- An ingredient record proves a product is authorized or clinically appropriate.
What This Article Does Not Support
This article does not support:
- Starting, stopping, substituting, diluting, combining, or continuing either antiseptic.
- Choosing an antiseptic for a patient, unit, procedure, wound, catheter, mucosal site, surgical field, infant, pregnant patient, or device.
- Product rankings, procurement decisions, formulary changes, or infection prevention policy changes.
- Treating in vitro findings, identity records, or labels as proof of clinical superiority.
- Moving evidence from one jurisdiction, formulation, or endpoint into another without source review.
Sources And Review
This article was editorially reviewed for source traceability on 2026-05-07. Key source categories included evidence reviews, ICU bathing studies, susceptibility research, CDC guidance, NICE and WHO surgical-site guidance, a DailyMed chlorhexidine product label, and an FDA substance identity record. Editorial review is not medical advice, regulatory advice, or a clinical recommendation.