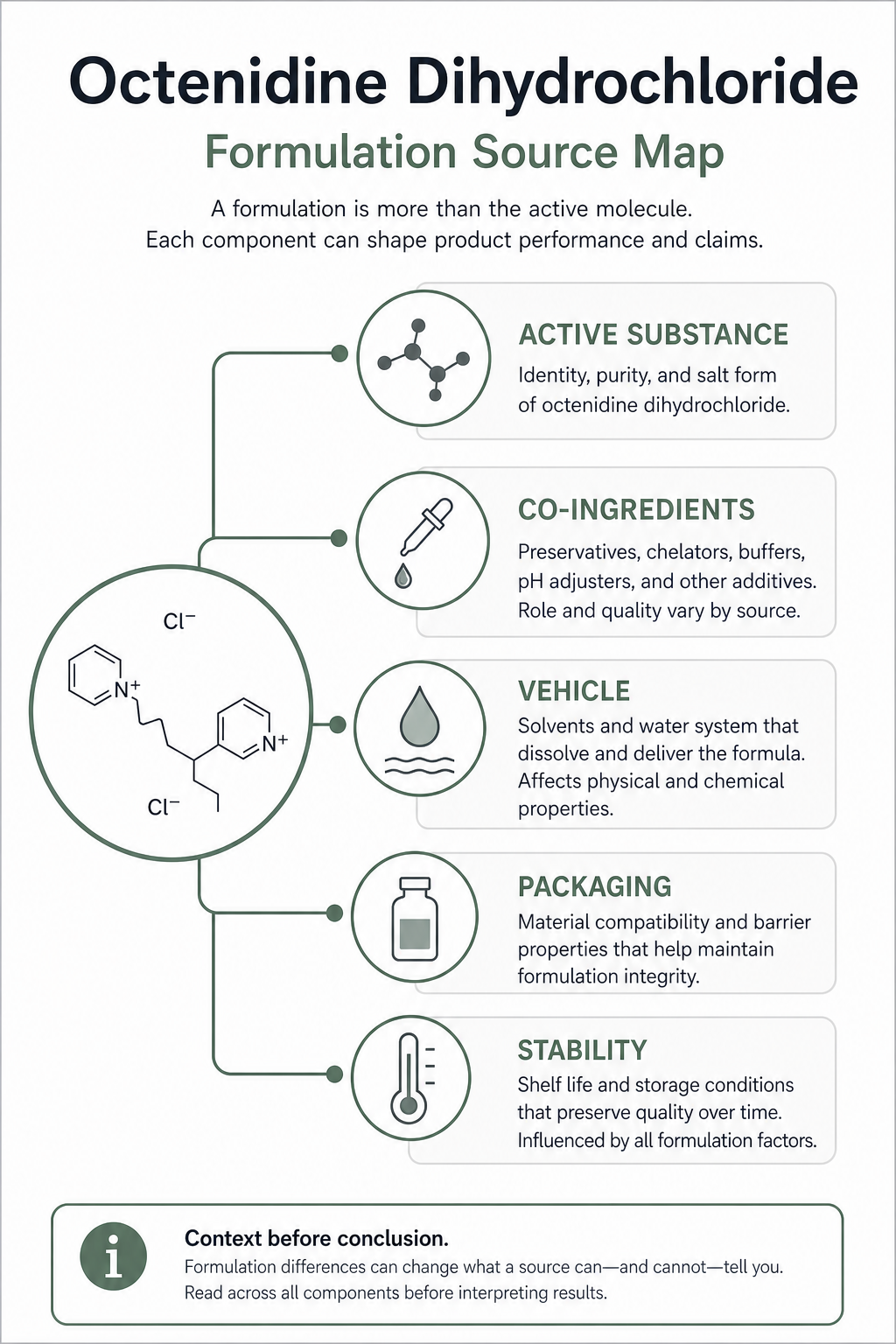
This article explains how industry readers can keep octenidine dihydrochloride formulation statements inside the documents that actually support them.
It is not formulation advice, regulatory advice, legal advice, procurement guidance, import guidance, supplier qualification, GMP assessment, product approval mapping, or a product-substitution guide. It does not recommend products, vendors, excipients, concentrations, vehicles, packages, or compatibility decisions.
Quick Position
Octenidine identity records and finished-product documents answer different questions. FDA GSRS lists OCTENIDINE HYDROCHLORIDE with UNII U84956NU4B, formula C36H62N4.2ClH, and synonym mappings that include OCTENIDINE DIHYDROCHLORIDE. GSRS also states that UNII availability is based on scientific identity characteristics and does not imply regulatory review or approval.
PubChem CID 51166 records octenidine hydrochloride with molecular formula C36H64Cl2N4, molecular weight 623.8, and InChIKey SMGTYJPMKXNQFY-UHFFFAOYSA-N. ECHA substance information associates octenidine dihydrochloride with EC/List number 274-861-8 and CAS number 70775-75-6. Those records are useful for identity and synonym checks; they do not answer which vehicle, co-ingredients, concentration, packaging material, stability program, label, or compatibility claim applies to a finished product.
Finished-product sources are narrower. The Australian TGA ARTG record for OCTENISEPT octenidine hydrochloride 1 mg/mL and phenoxyethanol 20 mg/mL solution bottle lists a specific ARTG ID, product name, registration type, sponsor, ingredients, licence category, and licence status. That supports statements about that Australian product record as checked on 2026-05-07. It should not be rewritten as a global formulation rule, a clinical recommendation, a supplier-quality claim, or proof that another octenidine-containing product has the same composition, label, packaging, warnings, or status.
What Formulation Statements Need
A formulation statement should name the exact object being discussed. “Octenidine dihydrochloride” can mean an identity record, an active ingredient in a product record, a salt form in an analytical method, or a component in a finished product. Those are different source questions.
For industry documentation, common formulation questions include:
- Active-substance identity: supported by official substance records such as GSRS, PubChem, and ECHA.
- Co-ingredient identity: supported by the product label, product information, formulation record, or inactive-ingredient source that actually lists the co-ingredient.
- Concentration or strength: supported by a product label, regulator product record, validated method result, certificate, or batch document for the exact product and version.
- Vehicle or dosage form: supported by product-specific labeling, product information, or formulation documents.
- Stability and storage: supported by product-specific stability data, label storage conditions, or stability guidance only within its scope.
- Packaging or material contact: supported by container-closure, primary-packaging, material-compatibility, extractables/leachables, or packaging records for the exact product or material.
- Compatibility: supported only by the exact compatibility study, label statement, material test, analytical result, or product document that answers the compatibility question being asked.
FDA’s inactive-ingredient database pages are a useful terminology boundary for U.S. drug products because they distinguish active ingredients from inactive ingredients in final dosage forms. They do not make an excipient appropriate for every octenidine product, route, body site, concentration, or jurisdiction.
Compatibility Means Several Different Things
“Compatibility” should not stand alone. It can mean chemical compatibility between active substance and co-ingredients, physical compatibility in a vehicle, analytical compatibility with a sample matrix, packaging or material compatibility, stability under storage conditions, photostability, or compatibility with a product’s labeled use context.
ICH Q1A(R2), as published by EMA, defines a stability data package for new drug substances and drug products that is sufficient for a registration application within ICH regions. ICH Q1B addresses photostability information for new active substances and associated medicinal products. Those guidelines help explain why stability and light-exposure statements are data-package questions, but they do not create an octenidine-specific shelf life, storage instruction, or compatibility claim.
Packaging and material statements are also scope-bound. EMA’s plastic immediate packaging materials guideline applies to human and veterinary medicines and covers specific requirements for plastic immediate packaging materials for active substances or medicinal products; it does not cover every container-closure property or every material type. FDA’s container-closure questions-and-answers guidance discusses container-closure suitability as something supported by data for the relevant storage or shipping context. Neither source should be used as a shortcut for saying that a particular octenidine formulation is compatible with a particular bottle, closure, applicator, wipe substrate, device surface, or use environment.
Product Registers And Labels
EMA’s national-registers page explains that EU and EEA national medicine registers contain information on medicines authorised in those countries, including links for the summary of product characteristics and package leaflet, and that this complements information on EMA’s own website. That is a jurisdiction boundary.
The EMA PSUSA page for octenidine dihydrochloride and phenoxyethanol is a pharmacovigilance procedure page for related nationally authorised products in the EU. It is useful for checking the active-substance combination and the documents attached to that procedure. It is not a global product list, a formulation equivalence table, a compatibility source for materials, or a reason to treat products from different countries as interchangeable.
When a product label or register gives concentration, co-ingredient, dosage-form, storage, or warning language, the narrow source-backed statement should identify the product, jurisdiction, document type, and date checked. A label can support what that label says about that product version. It cannot support a different product, a different package, a different body site, a different country label, or an unstated compatibility claim.
Documents That Often Get Mixed Up
Official substance records answer identity questions. Finished-product composition and compatibility need other sources.
A product label or regulator product record speaks for one product. It is not evidence that another product shares that formulation, approval status, clinical performance, or supplier quality.
A safety data sheet covers hazard communication and handling for the exact substance or mixture and document version. Body-site suitability, clinical safety, effectiveness, market authorization, formulation equivalence, and compatibility with other products or materials are different questions answered by different documents.
Stability reports, packaging studies, analytical reports, and certificates cover the tests, conditions, materials, batches, methods, acceptance criteria, and dates inside their own scope, when the documents are authentic and applicable. On their own, they do not prove GMP status, legal marketability, supplier qualification, product release suitability, or clinical value.
Peer-reviewed formulation and analytical studies cover the matrix, method, analyte, concentration range, product type, and endpoint actually studied. Pulling those results into a product-compatibility, clinical-performance, or interchangeability claim requires a source that directly supports it.
Writing Narrow Claims
Safer industry copy uses document-specific language:
- FDA GSRS lists OCTENIDINE HYDROCHLORIDE with UNII U84956NU4B and synonym mappings that include OCTENIDINE DIHYDROCHLORIDE.
- PubChem CID 51166 records octenidine hydrochloride identifiers that can help cross-check synonym clusters.
- ECHA associates octenidine dihydrochloride with EC/List number 274-861-8 and CAS number 70775-75-6.
- The TGA ARTG record checked on 2026-05-07 lists a named Australian product record with octenidine hydrochloride and phenoxyethanol as ingredients.
- EMA national registers are sources for country-specific medicine information and linked product documents, not a substitute for checking the current national record.
- A stability, packaging, material, analytical, or compatibility statement should name the exact product, concentration, material or matrix, condition, document version, and date.
Statements that need stronger or different sourcing include product approval outside the cited jurisdiction, market availability, legal classification, clinical safety, clinical effectiveness, supplier quality, GMP status, compendial compliance, batch release, product interchangeability, material compatibility, use-site suitability, and any recommendation about formulation selection.
Sources And Review
This article was editorially reviewed for source traceability on 2026-05-07. Key source categories included FDA GSRS, PubChem, and ECHA for identity; TGA and EMA pages for product-register and jurisdiction context; FDA inactive-ingredient pages for ingredient terminology; and ICH, EMA, and FDA quality guidance for stability, photostability, packaging, and container-closure source boundaries.
Editorial review is not medical advice, regulatory advice, legal advice, formulation advice, procurement advice, supplier qualification, GMP assessment, or a clinical recommendation.