The grocery aisle, the pharmacy shelf, and the cleaning cupboard all use words that sound similar. Antiseptic, antibiotic, disinfectant, sanitizer, preservative, wound cleanser. They are not the same thing, and the differences matter the moment you pick something up to use.

Quick Difference
The categories share words but not jobs.
An antiseptic works on living tissue. FDA describes over-the-counter consumer antiseptics as wash products like antibacterial soaps and rub products like hand sanitizers and antiseptic wipes. The agency separates consumer antiseptics from health care antiseptics because the settings and target populations differ. Octenidine sits inside the antiseptic family.
An antibiotic is a medicine that treats some bacterial infections. FDA tells consumers that antibiotics treat only some infections caused by bacteria and do not treat viral infections. That is a different category from a label-reading question about an antiseptic.
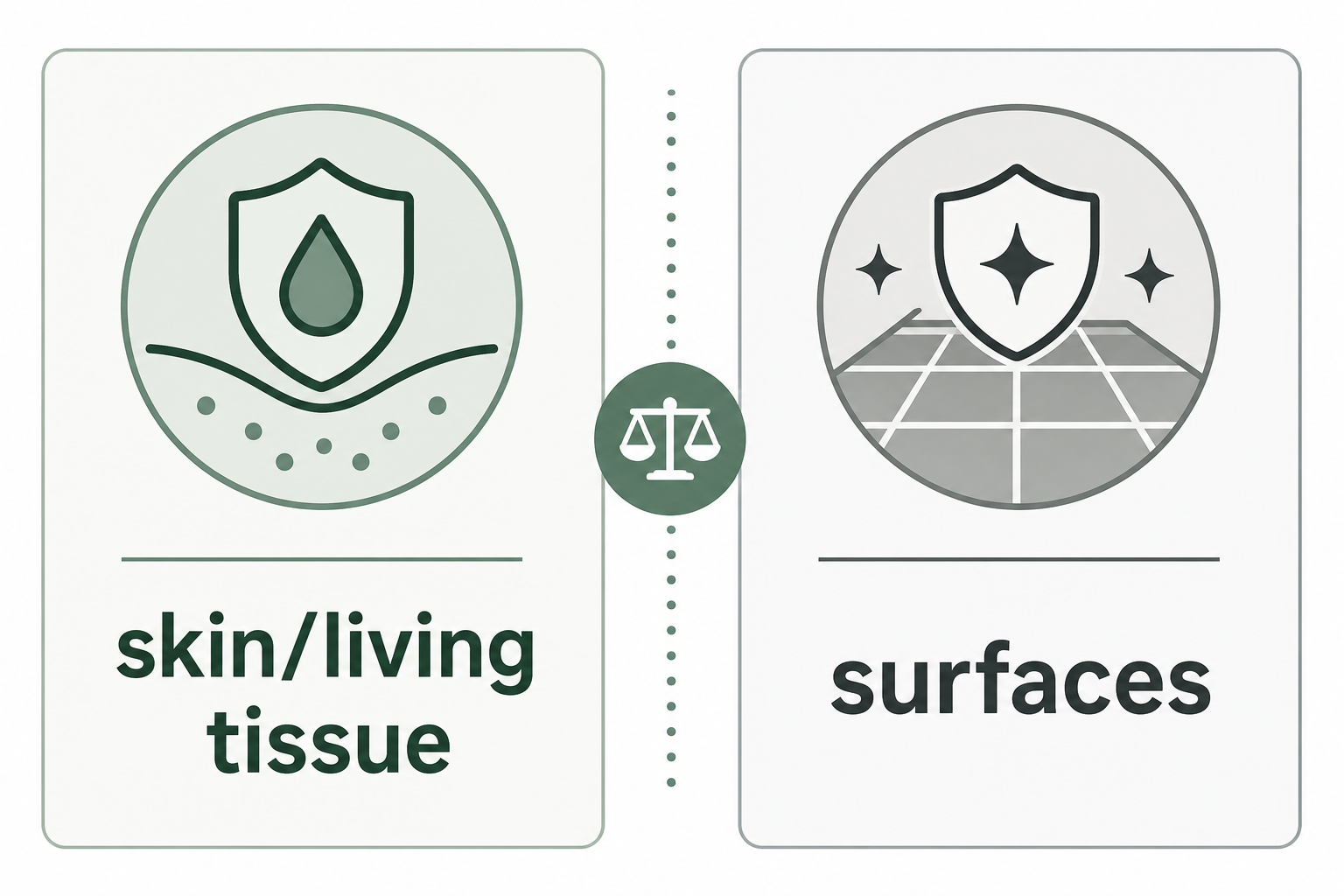
Disinfectants and sanitizers are usually about surfaces, not skin. EPA explains that products used to kill viruses and bacteria on surfaces are registered as antimicrobial pesticides, and that sanitizers and disinfectants are surface-product categories. CDC’s infection-control definitions also separate antiseptics for living tissue and skin from disinfectants for inanimate objects.
Preservatives are not the same as an antiseptic. FDA explains that preservation systems can help keep microorganisms from growing in cosmetic products. The job is to protect the product itself, not the user’s skin.
Wound cleansers are a small category with a lot of internal variation. Some are sterile saline or sterile water medical products; some are first-aid antiseptic solutions with an active ingredient like benzalkonium chloride. The label is what separates them.

Where Octenidine Sits
Inside the antiseptic family, octenidine is one of several familiar names alongside chlorhexidine, povidone-iodine, polyhexanide, and benzalkonium chloride. Each of those ingredients lives across a different label slate.
A few things make octenidine recognisable in this group. It pairs with phenoxyethanol in the most familiar EU products, which gives an aqueous formulation suitable for wound and mucous-membrane antisepsis under those national authorisations. It also pairs with alcohol in pre-procedure skin antiseptic products like octeniderm. And in oral and oromucosal labels, octenidine-based mouth rinses do not typically carry the chlorhexidine-style staining warning.
None of that turns octenidine into a default answer. It does mean octenidine has a label profile worth recognising on its own, separate from chlorhexidine, povidone-iodine, or any other antiseptic.
| Skin and tissue | Surfaces | |
|---|---|---|
| Examples | Antiseptic skin solutions, hand sanitizers, surgical skin preps, mouth rinses, wound antiseptics | Disinfectants, sanitizers, surface wipes, cleaning sprays |
| What they touch | Skin, mucous membranes, wound surfaces, oral cavity in a labelled context | Counters, instruments, floors, environmental surfaces |
| Regulatory home (U.S.) | FDA — drug or topical antiseptic categories | EPA — antimicrobial pesticide registrations |
| What goes wrong with confusion CDC, EPA, TGA | Surface chemistry on skin can injure tissue; skin antiseptics do not necessarily disinfect a surface | Surface disinfectants on skin can be harmful and are not approved for skin use |
Surface Products Are Not Skin Products
EPA’s consumer-facing surface page distinguishes cleaning, sanitizing, disinfecting, and hand sanitizer. EPA describes cleaning as removing dirt and organic matter, sanitizing as killing bacteria on surfaces, disinfecting as killing viruses and bacteria on surfaces, and hand sanitizer as a skin product regulated by FDA.
TGA’s consumer disinfectant page is direct about the boundary: disinfectants are designed for surfaces, are not to be taken internally, and are not for use on skin. CDC also notes that disinfectants are not used for skin antisepsis because they can injure skin and other tissues.
That boundary applies any time a label uses words like antimicrobial, disinfectant, sanitizer, cleaner, wipe, or spray. Similar words do not make products interchangeable.
Antibiotics Are A Different Conversation
Antibiotics are medicines for some bacterial infections, and FDA recommends talking with a healthcare professional when symptoms suggest infection. Antiseptic label language is not a substitute for diagnosis, antibiotic decisions, or care for symptoms that need a clinician.
The reverse is also true. A general statement about antibiotics does not tell you how to use an antiseptic. Keep the questions separate. Antibiotics are about diagnosed or suspected infections and professional medication decisions. Antiseptic products are about a labelled finished product, an intended use, warnings, and a local regulator.
Preservative Means The Product Is Being Protected
A preservative on a cosmetic ingredient list is doing a different job. FDA explains that cosmetic products can become harmful if contaminated with harmful microorganisms, and identifies ineffective preservation systems as one way that contamination can become a problem. The preservative protects the product, not the user.
FDA also notes that products in the health and beauty aisle can fall into different legal categories. Some are cosmetics, some are drugs, some are both, and some are medical devices. Intended use and claims drive the legal category.
That is why an ingredient list alone is not enough. A preservative name on a cosmetic label does not turn the product into an antiseptic, an antiseptic active ingredient on a drug label does not turn the product into a surface disinfectant, and a familiar chemical name cannot replace the product’s intended-use statement and warnings.
Wound Cleansers Need Extra Care
“Wound cleanser” sounds simple. Labels and product types vary a lot.
FDA has described sterile saline and sterile water medical products as water-based medical products used for irrigation or flushing of wounds or medical tubing. A DailyMed label for one wound wash antiseptic saline product, by contrast, lists benzalkonium chloride as the OTC active ingredient and describes first-aid antiseptic use for minor cuts, wounds, and scrapes.
Those examples show why the exact label matters. One wound-cleansing product may be a sterile saline or sterile water medical product, another may include an antiseptic active ingredient, and another may have different warnings or a different intended setting. The article does not decide which product belongs in any one situation.
Wound questions, especially those involving deep wounds, burns, surgical sites, chronic wounds, device-related care, infants, pregnancy, breastfeeding, allergies, immune compromise, diabetes, circulation problems, worsening symptoms, or unclear directions, belong with a qualified professional who can review the exact label alongside the situation.

A Safer Way To Read The Words
When category words blur together, the exact product label is the way back to clarity.
- What does the label call the product?
- What active ingredient or ingredient list is shown?
- What intended use, body site, surface, or setting is named?
- What warnings, age limits, allergy language, storage details, expiry, lot or batch code, and contact details are present?
- Does the label say to ask a doctor, pharmacist, or other professional?
FDA’s OTC Drug Facts label page and TGA’s medicine-label page both show why these details matter. They are label-reading sources, not product-selection tools.
Common questions
Is octenidine an antibiotic?
No. Octenidine is a topical antiseptic that acts on microorganisms on skin or mucous-membrane surfaces. Antibiotics are medicines for bacterial infections from the inside. Different categories, different jobs.
Can I use a surface disinfectant on a small cut?
No. Surface disinfectants are not for skin and can damage tissue. EPA, TGA, and CDC sources are explicit about this. Use a wound-cleansing product or antiseptic that is labelled for skin or wound use.
Is a hand sanitizer the same as a skin antiseptic?
FDA classifies hand sanitizers within the topical antiseptic category, but a hand sanitizer is not a wound-cleansing product or a surgical-skin prep. The label tells you what each one is for.
If a cosmetic ingredient list shows an antiseptic, is the product an antiseptic?
Not necessarily. A preservative can be present to protect the product itself. The intended use and claims on the label decide the legal category, not the ingredient list alone.
Why is octenidine treated as an antiseptic in some products and as a preservative in others?
The same ingredient can play different roles depending on the formulation and the label claims. In an aqueous skin and mucous-membrane antiseptic, octenidine is the active ingredient. In a wound irrigation gel that lists octenidine as a preservative, the role is to protect the product. The label decides which one applies.
Related Reading
For ingredient basics, read What octenidine is, in plain English. For a side-by-side label comparison with chlorhexidine, see Octenidine vs chlorhexidine: what their labels tell you. For product names and label families, see The octenidine product family, in plain English.
Sources And Review
Last reviewed: 2026-05-07. The references behind this article include FDA consumer antiseptic and antibiotic pages, CDC infection-control terminology, EPA and TGA surface-disinfectant resources, FDA cosmetic and medical-device context, a DailyMed product-label example, consumer label-reading pages, and U.S. poison-control resources. This page is editorial and is not medical advice, regulatory advice, emergency care, or a product recommendation.